- Anatomical terminology
- Skeletal system
- Joints
- Muscles
- Heart
- Blood vessels
- Lymphatic system
- Nervous system
- Respiratory system
- Digestive system
- Urinary system
- Female reproductive system
- Male reproductive system
- Endocrine glands
- Eye
- Ear
Adrenal glands
The adrenal glands (also known as suprarenal glands, singular in Latin: glandula suprarenalis) are two small endocrine organs located in the abdomen on top of each kidney. The adrenal glands regulate metabolism, immune system, blood pressure, and even response to stress by synthesizing and secreting steroid hormones in the bloodstream. Each adrenal gland have two main parts - adrenal cortex and medulla, each producing different hormones.
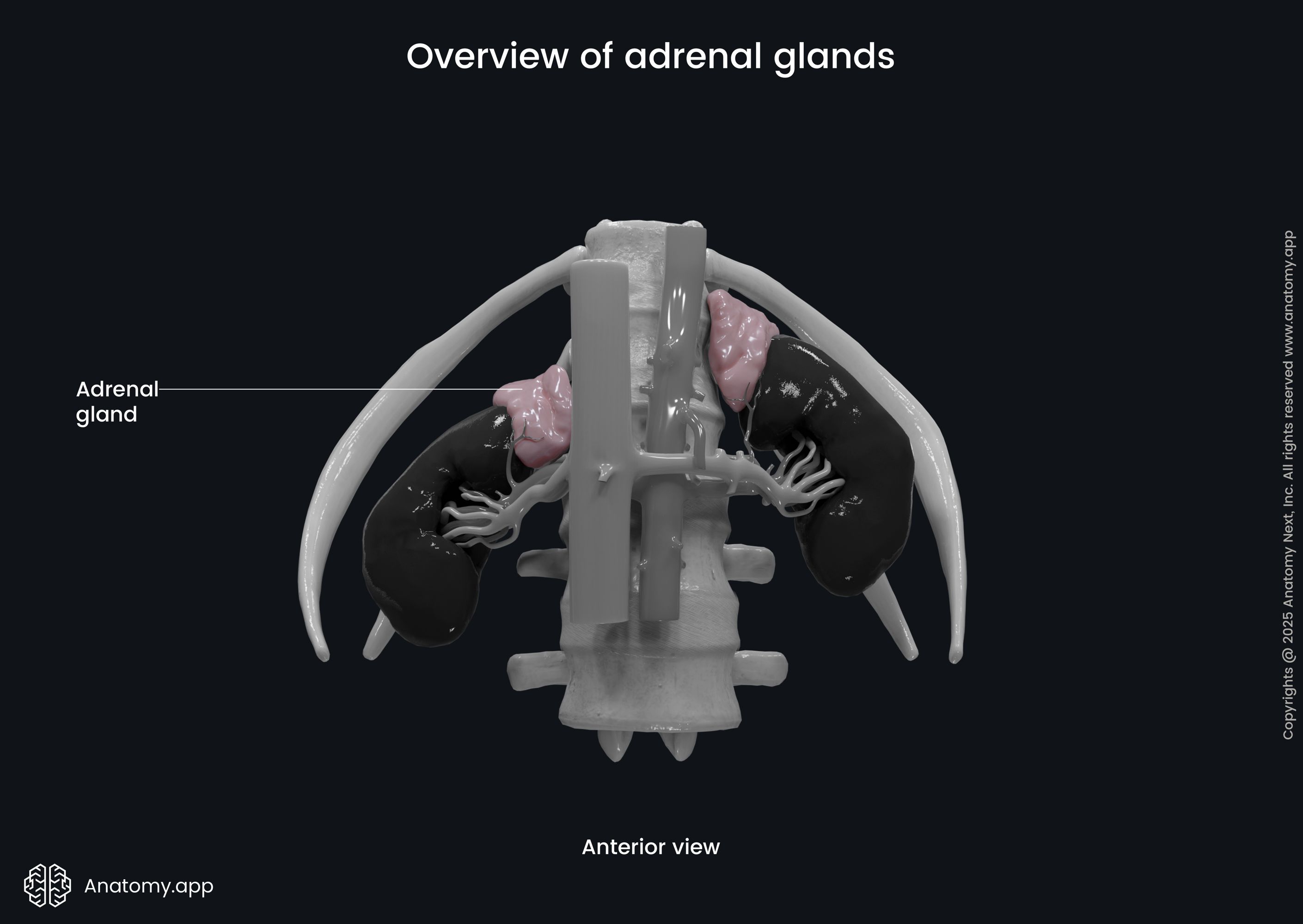
Adrenal gland anatomy
The adrenal glands are located at the posterior part of the abdomen. The peritoneum covers the glands from the anterior side, making them retroperitoneal organs. The adrenal glands lie on the top of the kidneys and are slightly medial to them. The color of the adrenal glands is yellowish. The right and left glands have different shapes. The right one is pyramid-shaped, while the left adrenal gland is semilunar-shaped and a bit larger. The weight of each gland is around 5 grams (0.18 ounce), length - 5 cm (2 inches), width - 3 cm (1.2 inches), and thickness - around 1 cm (0.4 inch).
Each gland is surrounded by a fatty capsule, and it lies within the perinephric or renal fascia together with the related kidney. The adrenal glands are separated from the kidneys by perirenal fat. The glands are located directly under the diaphragm. To the anterior side of the right adrenal gland are the inferior vena cava and right lobe of the liver, while posteriorly lies the right crus of the diaphragm. The stomach, pancreas, and spleen are located in front of the left adrenal gland, while the left crus of the diaphragm is behind the left gland.
Parts of adrenal gland
Each adrenal gland consists of two parts: outer part known as the adrenal cortex, and inner part - adrenal medulla. Both parts differ in their structure and function, and produced hormones. They also differ in their embryonic development, so the adrenal cortex and medulla can actually be considered as two separate endocrine glands.
Adrenal cortex
The adrenal cortex is the outer part of the adrenal gland, and it is also the largest part. It has a yellowish color. The cortex totally encloses the adrenal medulla. During the embryonic period, the cortex is derived from the embryonic mesoderm.
Cells of the adrenal cortex are classified into three types that produce specific hormones, and they are arranged in three layers or zones. Each zone therefore is responsible for producing and secreting specific hormone groups. From the outermost to innermost, the layers of the adrenal cortex are as follows:
- Zona glomerulosa - responsible for production of mineralcorticoids, mainly aldosterone;
- Zona fasciculata - produces glucocorticoids, for example, cortisol;
- Zona reticularis - produces androgens, for example, androstenedione - the precursor of testosterone.
Zona glomerulosa
The zona glomerulosa is the outer part of the adrenal cortex. This layer lies immediately below the fatty capsule surrounding the adrenal glands. The cells within the zona glomerulosa are arranged in clusters and resemble balls of wool. These cell groups are separated from each other with connective tissue. The zona glomerulosa is only five to seven cell layers thick, but it depends on its functional state. For example, an individual with chronic sodium deprivation will have a larger zona glomerulosa than one with a normal sodium intake.
The main cells of the zona glomerulosa have steroid-secreting cell characteristics with lipid droplets and large numbers of mitochondria. Connective tissues of this layer have many capillaries, so the secreted hormones can get directly into the bloodstream. The zona glomerulosa is responsible for secreting mineralcorticoids, mainly aldosterone. Aldosterone plays an important part in the regulation of arterial blood pressure. It affects kidneys to conserve salt and water.
Zona fasciculata
The middle zone of the adrenal cortex is called zona fasciculata. It is much thicker than the other two layers, accounting for approximately 80% of the adrenal cortex. The zona fasciculata consists of vacuolated cells that contain lipid droplets, abundant mitochondria, and a smooth endoplasmic reticulum. Like the cells of zona glomerulosa, these cells also have steroid-secreting characteristics.
The cells of the zona fasciculata are arranged in parallel radial cords that resemble long strings or bundles of sticks (fasciculi), stretching from the zona glomerulosa down in the direction of the adrenal medulla. The zona fasciculata is responsible for secreting glicocorticoids, including cortisol and corticosterone. These hormones affect metabolism, increase blood glucose levels, and take part in regulating the immune system. Another glicocorticoid, 11-deoxycorticosterone, is also secreted by the cells of the zona fasciculata. Zona fasciculata also secretes adrenal androgens, but much less than zona reticularis.
Zona reticularis
The zona reticularis is the most inner part of the adrenal cortex, lying between the zona fasciculata and adrenal medulla. It consists of cells forming a loose network. This layer also contains steroid-secreting cells with lipid droplets and many mitochondria. The zona reticularis is mainly responsible for secreting adrenal androgens (sex hormones), such as the dehydroepiandrosterone (DHEA), DHEA sulfate, and androstenedione. The zona reticularis also produces some glucocorticoids but in smaller amounts than zona fasciculata.
Adrenal medulla
The adrenal medulla can be considered as an interface between the endocrine and nervous systems, because it has characteristics of both systems. The medulla is the innermost part of each adrenal gland. It is surrounded by the adrenal cortex and is thinner compared to the cortex. Also, the adrenal medulla looks darker than the cortex. The adrenal medulla contains chromaffin cells that are brownish in color.
The cells of the adrenal medulla can be easily differentiated from the cells in the adrenal cortex, as the chromaffin cells do not have lipid droplets but have many secretory granules (therefore, the histological staining differs for both cells). The chromaffin cells are connected to the sympathetic part of the autonomic nervous system. These cells are actually modified postganglionic neurons with preganglionic autonomic fibers leading to them directly from the central nervous system.
The chromaffin cells produce catecholamines, including epinephrine (adrenaline), norepinephrine (noradrenaline), and dopamine. The adrenal medulla is responsible for around 20% of norepinephrine and 80% of epinephrine production. As the medulla has many capillaries, these hormones are secreted directly into the bloodstreem through the capillaries.
Note that even though catecholamines also work as neurotransmitters within the nervous system, here they are released into the bloodstream instead of the synaptic cleft. The hormones produced by the adrenal medulla work more rapidly and are short-lived compared to other hormones. Because the adrenal medulla is formed by neural tissue with direct neural input and produces substances that activate sympathetic nervous system, it actually functions like a modified sympathetic ganglion.
Neurovascular supply of adrenal glands
The adrenal glands are highly vascularized because they release hormones directly into the bloodstream. Innervation and neural input are interesting as the adrenal medulla is partly sympathetic ganglion itself.
Arterial blood supply
The adrenal glands have one of the most significant blood supply rates per gram of tissue compared to other organs in the human body. Around 60 small arteries enter each adrenal gland. Each gland is supplied by the following three main arteries:
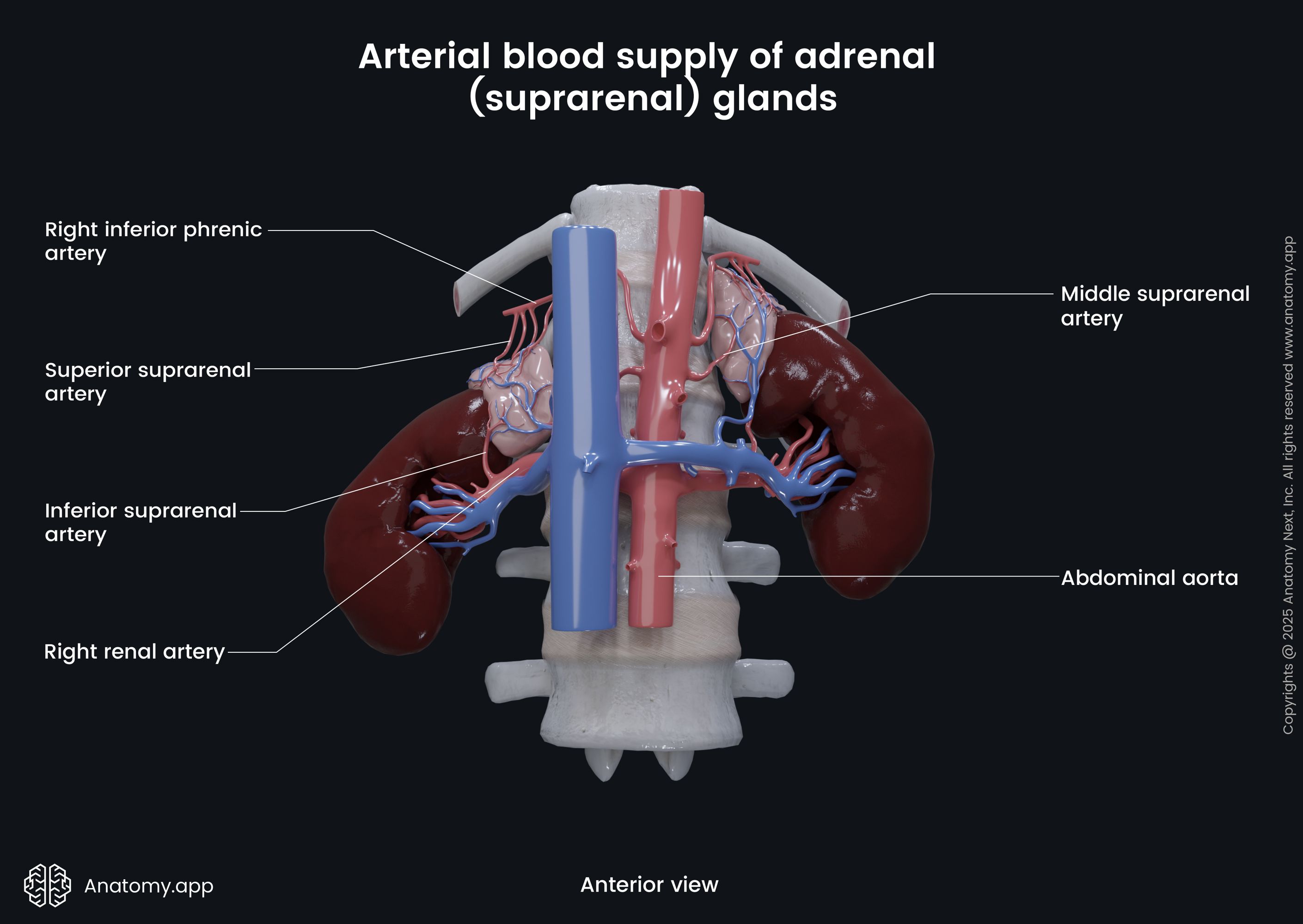
The superior suprarenal artery is a branch of the inferior phrenic artery. The inferior phrenic arteries are vessels that arise from the anterior surface of the abdominal aorta. At the upper medial part of the adrenal glands, the inferior phrenic arteries branch into the superior suprarenal arteries.
The middle suprarenal artery branches off directly from the abdominal aorta. The middle suprarenal arteries go horizontally to each side of the aorta and enter the adrenal glands at their lower medial parts.
The inferior suprarenal artery is a branch of the renal artery, which itself arises from the abdominal aorta. The left inferior suprarenal artery goes vertically and cranially to the left adrenal gland, but the right artery passes more horizontally towards the right gland.
Venous drainage
Venous drainage from the adrenal glands occurs through the suprarenal veins, one for each gland. The right suprarenal vein goes horizontally and drains into the inferior vena cava. The left suprarenal vein passes vertically and drains into the left renal vein, but it can also flow into the left inferior phrenic vein.
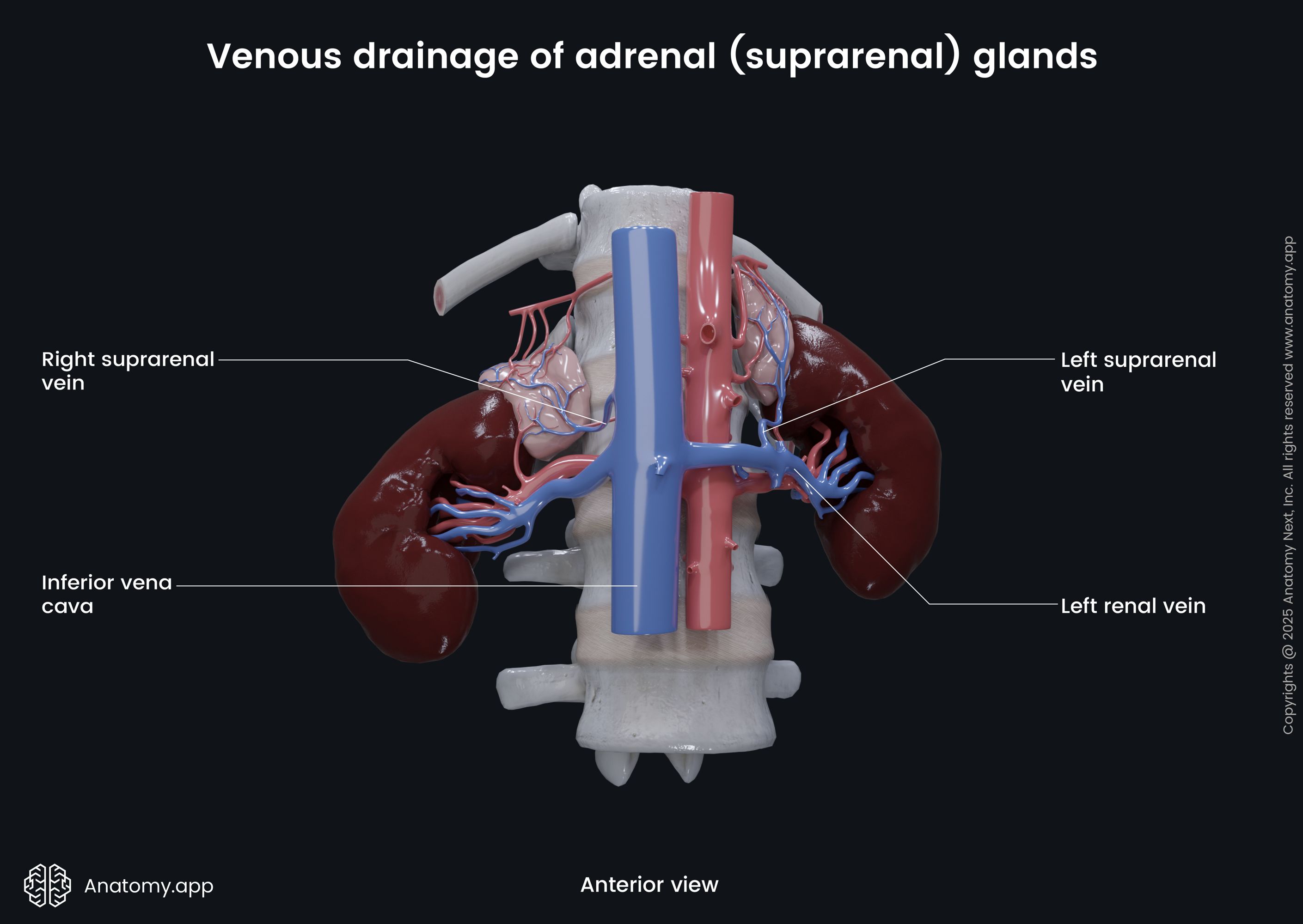
An unusual type of vein, the central adrenomedullary vein, is located in the adrenal medulla. Its main difference is in its structure, as the smooth muscles in tunica media are arranged differently than in other veins - in longitudinally oriented bundles.
Lymphatic drainage
The adrenal glands have two lymphatic plexuses, one in the medulla and one in the adrenal capsule. Most of the lymph drains into the lateral aortic lymph nodes and the para-aortic nodes. Smaller amounts of lymph drain into the thoracic duct or the posterior mediastinum.
Innervation and hormonal stimulation
The adrenal glands are regulated by neuronal and hormonal stimulation. The adrenal cortex is stimulated by the adrenocorticotropic hormone produced by the anterior pituitary gland. The adrenocorticotropic hormone stimulates the adrenal cortex to produce corticosteroids.
The adrenal glands are innervated by fibers of the sympathetic nervous system through the celiac ganglion and the greater splanchnic nerve. The adrenal medulla is supplied by the sympathetic preganglionic nerve fibers that arise from the spinal cord segments T5 - T8 and bypass the paravertebral sympathetic ganglion.
Afterward, the fibers form the greater splanchnic nerve. Some of the nerve fibers will synapse at the celiac ganglion, and its post-synaptic fibers innervate the blood vessels that supply the adrenal glands. Other fibers will avoid celiac ganglion and go to the adrenal glands to innervate the chromaffin cells in the adrenal medulla. The chromaffin cells work as cells of a ganglion and give rise to post-ganglion fibers, which release neurotransmitters directly into the bloodstream.
Adrenal gland functions
The adrenal functions are based on the hormones the glands secrete. Each hormone has its specific functions, but they may overlap. The adrenal cortex and adrenal medulla secrete entirely different hormones. As mentioned before, the adrenal cortex produces steroid hormones: mineralocorticoids, glucocorticoids and androgens. The adrenal medulla, on the other hand, is responsible for production of catecholamines, which are essentially neurotransmitters.
Adrenal cortex hormones and their functions
Mineralocorticoids
Aldosterone is the primary mineralocorticoid produced by the adrenal cortex in the zona glomerulosa. Its leading role is in water and salt balance. Aldosterone is involved in regulating blood pressure, by influencing blood sodium and potassium levels. It happens through its participation in the renin-angiotensin-aldosterone system.
If the renal perfusion is reduced or the sodium levels in the blood are low, specific cells within the kidneys (cells of the juxtaglomerular apparatus) sense this change and in response release renin into the bloodstream. Renin is a substance that modifies the inactive angiotensinogen, produced by the liver, changing it into angiotensin I. Angiotensin I is a small protein that circulates through the blood and is again cleaved by an angiotensin-converting enzyme to form angiotensin II.
Angiotensin II stimulates cells within zona glomerulosa of the adrenal cortex to produce and secrete aldosterone. Aldosterone promotes sodium and water retention in the kidneys and potassium excretion, therefore, increasing sodium levels and decreasing potassium levels in the blood. By influencing sodium and potassium levels, aldosterone indirectly affects water retention or loss, blood pressure, and blood volume.
If baroreceptors sense decreased blood pressure, aldosterone is released that causes sodium and water retention leading to increased blood volume and blood pressure. If aldosterone is produced too much, it is known as hyperaldosteronism, while too little aldosterone is called hypoaldosteronism.
The hypothalamus regulates aldosterone levels through the hypothalamic-pituitary-adrenal (HPA) axis. If aldosterone level decreases, the hypothalamus produces corticotropin-releasing factor (CRF) that stimulates cells in the anterior part of the pituitary gland. In response, the pituitary gland releases adrenocorticotropic hormone (ACTH). ACTH activates formation of deoxycorticosterone that is a precursor of aldosterone, as well as stimulates release of other adrenal cortex hormones. Note that sympathetic nerves also stimulate aldosterone production. Pain, emotions, anxiety (experiences generated in the brain) increase aldosterone levels through sympathetic stimulation.
Glucocorticoids
As mentioned before, the zona fasciculata of the adrenal cortex is responsible for secreting glucocorticoids, such as cortisol, corticosterone, and 11-deoxycorticosterone. Cortisol release mostly depends on stress and low blood glucose levels. Cortisol stimulates gluconeogenesis to generate glucose from glycogen in the liver. Cortisol indirectly affects glycogenolysis, by breaking down of glycogen. Increased amounts of cortisol can start the breakdown of proteins.
Cortisol also takes part in regulating immune responses. It inhibits the release of substances that cause inflammation. Cortisol blocks the production of interleukin-12, interferon-gamma, interferon-alpha, and tumor necrosis factor-alpha. Indirectly cortisol stimulates osteoclasts that can lead to reduced bone formation. Cortisol also takes part in regulating electrolyte balance by increasing sodium absorption through the small intestine and increasing potassium excretion. Interestingly, cortisol works together with adrenaline to form short-term memories.
The hypothalamus also regulates the release of cortisol through the hypothalamic-pituitary-adrenal (HPA) axis. The hypothalamus secretes the corticotropin-releasing factor (CRF) that stimulates the anterior pituitary gland to secrete the adrenocorticotropic hormone (ACTH). ACTH acts upon the cells within the adrenal cortex to release cortisol and other glucocorticoids, and also mineralocorticoids and androgens.
In the human body, corticosterone has only weak potency of glucocorticoids and mineralocorticoids, and is important mainly in the process of creating aldosterone from pregnenolone (its precursor). Corticosterone may also have effects on memory. It may take part long-term memories associated with fear and stress, as it levels increase when people reactivate fearful or stressful memories.
11-deoxycorticosterone (DOC) is actually a mineralocorticoid with a weaker activity than aldosterone. However, it also has glucocorticoid activity. DOC is a precursor for the production of aldosterone. DOC participates in regulating electrolyte balance by stimulating reabsorption of sodium un excretion of potassium.
Androgens
Androgens secreted by zona reticularis include dehydroepiandrosterone, dehydroepiandrosterone sulfate, and androstenedione, as well as testosterone (less than 5% of the whole testosterone). Dehydroepiandrosterone (DHEA) acts as an intermediate in the biosynthesis of other sex hormones, including other androgens and estrogen. DHEA is a precursor to testosterone and dihydrotestosterone.
Together with other androgens, DHEA is responsible for androgenic effects during adrenarche, the "awakening of the adrenal glands" that happens before puberty around age 7 to 9 and induces changes affecting boys and girls around this time. These changes include axillary and pubic hair growth, adult-type odor, oiliness of skin and hairs, and mild acne. DHEA can also be converted into estradiol that provides estrogenic effects in some tissues, like the vagina. DHEA is also known to act upon some neurotransmitter receptors, like NMDA or GABAa receptors.
Dehydroepiandrosterone sulfate (DHEA-S) is another androgen produced by the zona reticularis. It can be turned into DHEA, which then can be transformed into testosterone and dihydrotestosterone. DHEA-S acts on the same neurotransmitter receptors mentioned before. Androstenedione is weak androgen, which takes part in the biosynthesis of estrogen and testosterone from DHEA. Androstenedione level increases during adrenarche. It also affects how children develop and understand sexual attraction.
Similarly to other adrenal cortex hormones, the production of androgens is also stimulated by the adrenocorticotropic hormone (ACTH) released by the anterior part of the pituitary gland. The ACTH production in the pituitary gland is stimulated by the corticotropin-releasing factor (CRF) produced by the hypothalamus. This interaction is known as the hypothalamus-pituitary-adrenal (HPA) axis.
Adrenal medulla hormones and their functions
The adrenal medulla produces catecholamines: epinephrine, norepinephrine, and dopamine. Cateholamines are neurotransmitters, however, the adrenal medulla release them directly into the bloodstream, therefore, they also act as hormones. Epinephrine (also called adrenaline) and norepinephrine (noradrenaline) are the main hormones produced by the adrenal medulla.
Both epinephrine and norepinephrine act on adrenergic receptors. However, epinephrine primarly affects the heart, while norepinephrine acts mostly on blood vessels. Epinephrine increases heart rate and contractility, induces relaxation of airways resulting in faster breathing, raises blood glucose levels, increases blood pressure by vasoconstriction, and increases physical performance and strength. Norepinephrine mainly increases blood pressure by vasoconstriction.
The adrenal medulla produces only small amounts of dopamine. Dopamine is a neurotransmitter that acts on the central nervous system and participates in creating the feelings of pleasure. It is released when an individual is doing anything that makes them happy. Dopamine boosts mood, as well as increases motivation and attention.
Adrenal gland disorders
Adrenal gland disorders are mostly characterized by hyper- or hypofunction of the gland leading to excess or too little production of adrenal hormones. Some of the most common disorders that affect adrenal cortex include Addison's disease (primary adrenal insufficiency), Cushing's syndrome, and congenital adrenal hyperplasia. A significant pathology affecting the adrenal medulla is a rare tumor originating from cells of the medulla, known as pheochromocytoma.
Addison's disease (adrenal insufficiency)
Addison's disease is the most common cause of primary adrenal insufficiency, however, it is a rare disease. Adrenal insufficiency is a state when the adrenal glands don't produce enough cortisol and aldosterone. Less common primary causes of adrenal insufficiency are adenoma of the adrenal gland, and congenital adrenal hyperplasia. Secondary causes include pathologies outside the adrenal glands, and may include problems of the pituitary gland or hypothalamus. Symptoms of adrenal insufficiency usually develop over time and include:
- Fatigue
- Weight loss
- Hyperpigmentation
- Low blood pressure
- Salt craving
- Abdominal pain
- Irritability
- Muscle or joint pain
- Low blood glucose levels
- Depression
- Body hair loss and irregular menstrual cycles (in women)
Treatment of adrenal insufficiency includes administration of additional glucocorticoids (cortisol) and aldosterone. If undiagnosed or untreated, it can lead to an adrenal crisis that is a life-threatening condition. Its symptoms include:
- Sudden pain in legs or lower back
- Confusion
- Low blood pressure
- Severe weakness
- Convulsions
- Fever
- Syncope
Blood tests may reveal hyperkalemia, hypercalcemia, hypoglycemia, hyponatremia. Adrenal crisis needs immediate emergency treatment. Treatment includes giving intravenous saline, hydrocortisone, and glucose.
Cushing's syndrome
Cushing's syndrome is a collection of symptoms caused by too much cortisol production. The most common cause is pituitary adenoma (a benign tumor) causing excessive secretion of adrenocorticotropic hormone (ACTH), and this condition is known as Cushing's disease. Cushing's syndrome may also develop after long-term usage of glucocorticoids, as it may be seen, for example, in patents who receive treatment for rheoumatoid arthritis. The symptoms of Cushing's syndrome include:
- Weight gain
- High blood pressure
- Irritability
- Excess hair growth in women
- Moon face
- Immunological function disorders
- Extra fat around the neck
- Irregular menstrual cycles
- Red face
- Poor concentration
If the cause is a pituitary adenoma, the first line of treatment is surgical resection of the tumor. Also, medications can be prescribed to control the production of cortisol. If it is caused by glucocorticoid medication, then the dosage may need to be decreased or alternative pharmacotherapy considered.
Congenital adrenal hyperplasia
Congenital adrenal hyperplasia is an autosomal recessive disorder with affected cortisol synthesis. It has many subtypes based on which of the five enzymes needed for the synthesis of cortisol is affected. The most common type is so-called classic adrenal hyperplasia. Symptoms of this disorder may include:
- Vomiting with following dehydration (inadequate mineralocorticoids)
- Too much androgens can cause:
- Excessive virilization
- Ambiguous genitalia in infants
- Early pubic hair in children
- Early or delayed puberty
- Irregular menstrual cycles
- Infertility
- Too little androgens and estrogens may cause:
- XY males to have female external genitalia
- Infertility, no development of sexual characteristics in females
The treatment is based on lowering or increasing hormone amounts. Hyperplasia can be reduced by giving enough glucocorticoids. Glucocorticoids can also help with the overproduction of androgens and mineralocorticoids. If the deficiency is in mineralocorticoids, then additional mineralocorticoids are given. If puberty is too late or not efficient, replacement treatment of testosterone or estrogen is needed.
Pheochromocytoma
Pheochromocytoma is a rare, benign tumor that arises from the chromaffin cells of the adrenal medulla. It causes the adrenal medulla to release tremendous amounts of catecholamines. The symptoms are caused by catecholamines, and include the following:
- High blood pressure
- Tremors
- Rapid heartbeat
- Panic attack symptoms
- Shortness of breath
- Headache
Due to excess cateholamines, pheochromocytoma can lead to complications, such as hypertensive crisis, myocardial ischemia, cardiomyopathy, arrythmias, stroke, or acute renal failure. Primary treatment of pheochromocytoma is surgical removal of the affected gland (adrenalectomy). In preparation for surgery, medications such as alpha-blockers are needed to decrease blood pressure. Beta-blockers may be prescribed to lower the heart rate.